
GTAC@MGI supports neonatology and clinical genetics teams with rapid whole genome sequencing for infants in intensive care. Our laboratory workflows are structured to deliver high quality sequencing data on a timeframe aligned with clinical decision making, in close partnership with the Department of Pathology & Immunology at WashU Medicine.
Critically ill infants often present with complex clinical findings that do not point toward a single diagnosis. Rapid whole genome sequencing can evaluate all major variant classes in one test and provide clarity when standard workups are unrevealing.
Congenital anomalies, seizures, metabolic instability, neurologic findings, or multiorgan involvement without a clear etiology.
Whole genome sequencing detects copy number changes, structural variants, and single gene disorders in one assay.
Published studies show rapid sequencing often informs medical management, guides additional testing, or clarifies prognosis.
rWGS follows a structured workflow that moves samples from receipt to sequencing in several days, enabling clinical teams to begin interpretation as soon as run outputs are available.
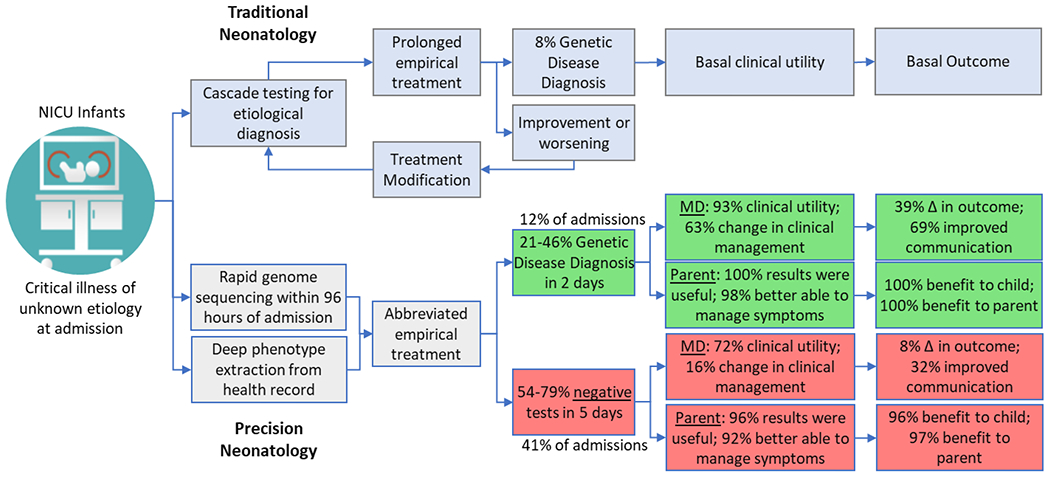
NICU rapid sequencing is a coordinated effort between GTAC@MGI and the Department of Pathology & Immunology. GTAC@MGI performs rapid whole genome sequencing and laboratory quality control. Clinical analysis, interpretation, and reporting are performed by the Department of Pathology & Immunology as part of standard clinical care.
rWGS is often informative for presentations that span multiple organ systems or do not fit a single diagnosis.
Unexplained acidosis, organ dysfunction, or recurrent decompensation.
Congenital anomalies, seizures, tone abnormalities, or brain imaging differences.
Situations where family history or recurrence risk informs diagnosis and management.
If you are coordinating NICU care and need rapid whole genome sequencing support, we can connect the right GTAC@MGI and Pathology teams and align on sample logistics and timing.
Sign up to receive updates from the McDonnell Genome Institute on new technologies, upcoming events, and occasional promotions on services that support your research.
By submitting this form you agree to receive email from the McDonnell Genome Institute at Washington University School of Medicine. You may unsubscribe at any time. Your information will not be sold or shared with outside organizations.